Reverse Osmosis
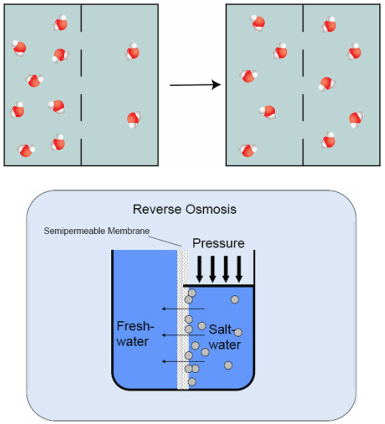
Osmosis is a natural process in which water passes through a semi-permeable membrane from a solution with a high salt concentration to a solution with a low salt concentration until dynamic equilibrium is established. In the state of dynamic equilibrium, the pressure over both sides of the membrane are.
Reverse Osmosis applies external pressure that is greater than the osmotic pressure of both solutions. This additional pressure forces the dissolved salt to be separated from the water.
Reverse osmosis processes are divided into three categories:
Reverse Osmosis applies external pressure that is greater than the osmotic pressure of both solutions. This additional pressure forces the dissolved salt to be separated from the water.
Reverse osmosis processes are divided into three categories:
- High-pressure reverse osmosis (5 – 10 MPa, as in seawater desalination)
- Low-pressure reverse osmosis (2 – 5 MPa, as in brackish water desalination)
- Nanofiltration (0.3 – 2 MPa, as in softening of drinking water)
